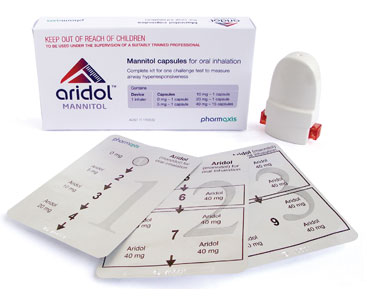
Aridol® in Australia
Aridol (mannitol) is manufactured by Pharmaxis Ltd (Frenchs Forest, Sydney, Australia) and is distributed in Australia by our distributor BTC Speciality Health Pty Ltd.
The mannitol challenge test (Aridol®) is an indirect osmotic bronchial challenge test that can be used to identify bronchial hyperresponsivesness, a key clinical feature of respiratory conditions such as asthma.
Aridol is manufactured by Pharmaxis Ltd (Frenchs Forest, Sydney Australia) and is available as a standardised test kit containing pre-filled mannitol capsules and a hand held dry powder inhaler.
During a mannitol challenge test, the subject inhales increasing doses of mannitol with their lung function (forced expiratory volume in one second- FEV1) measured after each dose to determine the level of bronchial hyperresponsiveness.
The mannitol challenge test should only be performed by a suitably trained healthcare professional. Please refer to the approved product information or summary of product characteristics for your country prior to performing this challenge test.
Please refer to the Approved Aridol Product Information(PI) and Consumer Medicine Information attached below.
For more information about Aridol in Australia please contact BTC Speciality Health Pty Ltd.
To Order
Email: SigmaHospitals@centralhs.com.au
Phone: 1800 646 991
Product Enquiries
Phone: Toll free number (24 hours): 1800 100 282
Email: queries@btchealth.com.au
Business hours – Monday to Friday (excluding public holidays) 9:00am – 5:00pm AEST
Medical Information and Adverse Events
Email: quality@btchealth.com.au.
Phone: 1800 100 282
Phone: +61 404 869 472
BTC Speciality Health Pty Ltd
Level 1, 10 Oxley Road
Hawthorn, VIC, 3124 Australia



 Australia
Australia
 Canada
Canada
 Chile
Chile
 Denmark
Denmark
 Ireland
Ireland
 Korea
Korea
 Norway
Norway
 Spain
Spain
 Sweden
Sweden
 United Kingdom
United Kingdom
 United States
United States